Medifuge & CCF Patent pending - Blood Phase Separator
Rotor Phase separator:
The Silfradent Medifuge MF200 is a dedicated medical plasma centrifuge. It is designed to prepare autologous concentrated growth factor (CGF) and CD 34 + cells using patient's own blood which is the ideal autologous source without the addition of exogenous substances. CGF represents a new generation of platelet that able to hold inside a higher concentration of autologous.
Growth factors are proteins which regulate the complex processes of wound healing and enhance the body's healing abilities without side-effects. The Medifuge prepares up to 8 test tubes of various levels of concentrate with active proteins in liquefied or Fibrin form to create membranes, glue or other particulars for tissue regeneration and bone augmentation in a short period of 12 minutes.
Medifuge MF200 Technical Specifications:
Product Features:
Rotor phase separator: Sifradent key points:
- 34+
- Morphogenetic cells
The Silfradent Medifuge MF200 is a dedicated medical plasma centrifuge. It is designed to prepare autologous concentrated growth factor (CGF) and CD 34 + cells using patient's own blood which is the ideal autologous source without the addition of exogenous substances. CGF represents a new generation of platelet that able to hold inside a higher concentration of autologous.
Growth factors are proteins which regulate the complex processes of wound healing and enhance the body's healing abilities without side-effects. The Medifuge prepares up to 8 test tubes of various levels of concentrate with active proteins in liquefied or Fibrin form to create membranes, glue or other particulars for tissue regeneration and bone augmentation in a short period of 12 minutes.
Medifuge MF200 Technical Specifications:
Product Features:
- This unique medical device allows for the use of up to 8 test tubes for the creation of CGF (fibrin).
- A micropocessor control system allows for the maintaining of a constant speed.
- The exception rotor system with self-ventilation protects the blood sample from heat exposure. The rotor-holding compartment, the closing door and the test tube-holding jackets guarantee biological safety in terms of bio-containment, in the event of test tube breakage.
- The test tube-holding jackets and rotor are built from thermal, antistatic material that is easy to clean, extract and sterilise in an autoclave at 135°.
- MEDIFUGE is equipped with a decontamination cycle with UVC reflected light. Cycle duration 5 minutes at 1,000 revs.
- The electronic control engine and its internal parts require no maintenance.
- Noise levels fall below the standards required and do not exceed 57 dBa.
Rotor phase separator: Sifradent key points:
- Rotor antistatic antimagnetic material (No influence on Fe in the red blood cells, minerals, heavy metals, decreases cancer-promoting substances as Aflatoxins, Cerium, Polonium, eliminates Clostridium)
- Differentiated acceleration
- Speed and fractional – alternating RCF (does not allow the platelet degranulation)
- Unique features: 4 different processes for obtaining in the blood separation: Serum, PPP, PRP, Concentrated Growth Factors, Red Blood, and fragments.
- High Concentrated Growth Factors are very important for the regeneration and repair of the tissue.
- 34+
- Morphogenetic cells
- Tubes-holders design for the positioning of the fragments in the bottom of the Tubes.
- Constant temperature maintained by self-ventilation control to avoid altering the natural process of sedimentation.
- Cycle Bleaching sterilizing= decontamination
- Rotor with alternate and controlled speed with an acceleration always under RCF300
- Cooling
|
Medifuge uses a program with the following characteristics:
30” acceleration phase 2’ at 2,700 rpm 4’ at 2,400 rpm 4’ at 2,700 rpm 3’ at 3,000 rpm 36” deceleration and stopped phases |
Features:
Capacity: 8 units (test tubes) Processing Time12 minutes Power Source: 230V +/- 10% 50/60 Hz Noise Level: Don't exceed 57 dB(A) Dimensions of Unit: 230w x 320L x 240H mm Weight: 9.4 Kg Country of Manufacture: Italy |
Medifuge Features:
Capacity: 8 units (test tubes)
Processing Time12 minutes
Power Source: 230V +/- 10% 50/60 Hz
Noise Level: Don't exceed 57 dB(A)
Dimensions of Unit: 230w x 320L x 240H mm
Weight: 9.4 Kg
Country of Manufacture: Italy
Capacity: 8 units (test tubes)
Processing Time12 minutes
Power Source: 230V +/- 10% 50/60 Hz
Noise Level: Don't exceed 57 dB(A)
Dimensions of Unit: 230w x 320L x 240H mm
Weight: 9.4 Kg
Country of Manufacture: Italy
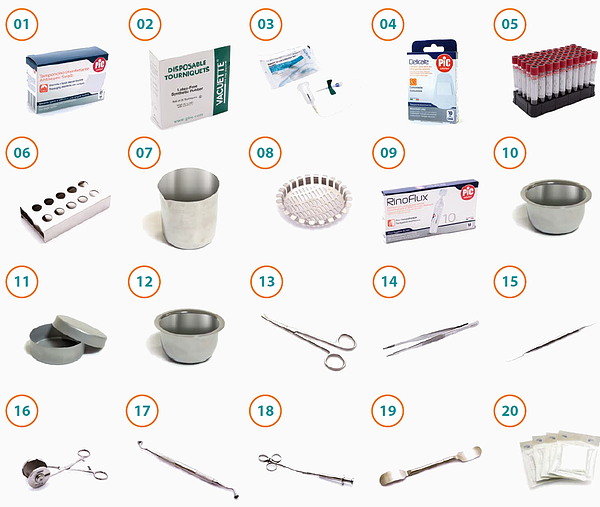
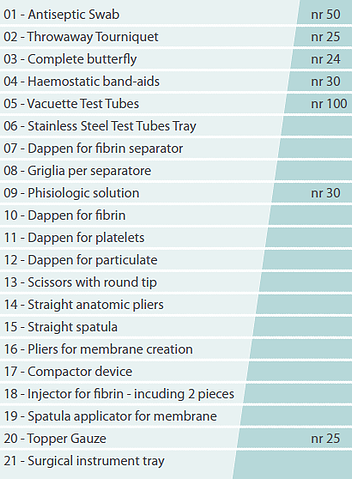
CCF General kit
The patent kit is equipped with the necessary for blood collection; specific happens to prevent any contamination risks and the required instrument to produce the fibrin membrane and insertion of the clot in the site.
The patent kit is equipped with the necessary for blood collection; specific happens to prevent any contamination risks and the required instrument to produce the fibrin membrane and insertion of the clot in the site.
CCF Cosmetic kit